ResearchHero eDiary for Clinical Trials
Empowering patient-centric research with seamless data collection, reminders, compliance visibility, and study-ready reporting.
Where innovation meets patient engagement
ResearchHero eDiary simplifies protocol-aligned data collection while prioritizing patient convenience, engagement, and compliance—supporting stronger study outcomes.
Key eDiary features
Seamless Integration
Effortlessly incorporate eDiary into study protocols and workflows.
Patient-Centric Design
Intuitive experience accessible from any internet-connected device.
Customizable Forms
Tailor questionnaires and form designs to study requirements.
Enhanced Engagement
Diverse form designs to encourage participation and accuracy.
Advantages of ResearchHero eDiary
- Effortless enrollment with accurate setup from day one.
- Patient convenience across devices and flexible schedules.
- Intuitive interface and form designs that support compliance.
- Improved engagement that strengthens data completeness.
Patient Journey with eDiary
Consent & Information Sharing
Patients review and consent to data sharing aligned with study requirements.
Profile & Study Details
Patients enter basic information and study-specific details provided by the clinic.
App Tutorial
Guided walkthrough of app features, surveys, and study information.
Daily Reminders
Patients configure timing/frequency of reminders based on their routine.
Demographics
Demographic inputs help support analysis and study insights.
Participant Information
Additional protocol-required information (history, lifestyle, meds, etc.).
Daily Questionnaires & Reminders
- Prompt submission of daily reports for consistent data collection.
- User-friendly interface designed for quick completion.
- Reminders to reduce missed or late entries.
- Real-time data capture with secure storage for immediate access.
- Seamless integration with patient routine to improve adherence.
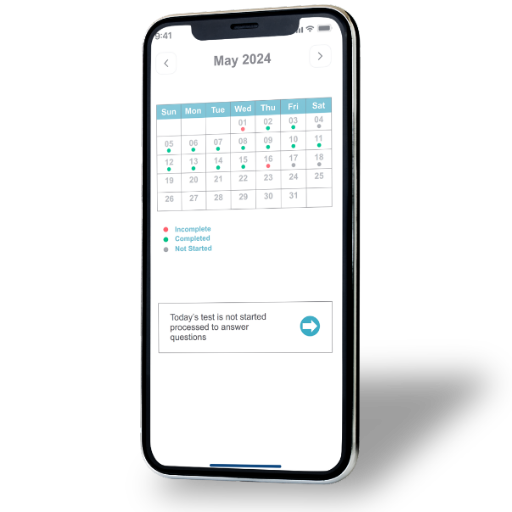
Data Recording & Analysis
- Secure and reliable data storage to support confidentiality and integrity.
- Comprehensive tracking of daily submissions for complete participation view.
- Calendar-style visibility of submission trends over time.
- Real-time access for review, monitoring, and analysis.
- Analytics support to extract actionable study insights.
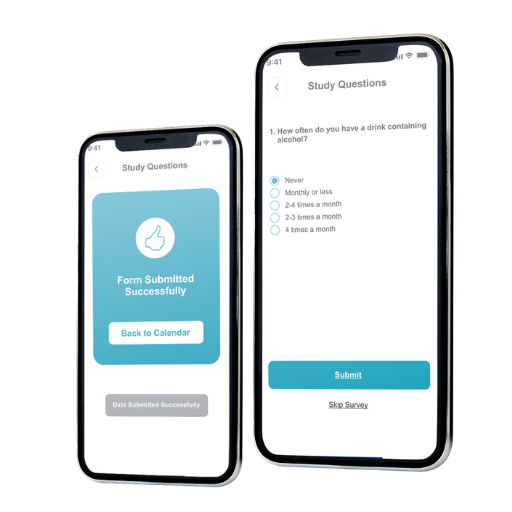
Alcohol Trials Module (Example)
Configure questionnaires to match alcohol study objectives and capture detailed drinking behavior with precision.
- Customize questions for frequency, beverage types, and consumption patterns.
- Flexible questionnaire design for nuanced behaviors and triggers.
- Support research precision by aligning prompts to study parameters.
Automated Intake Calculation
Automated calculation of daily alcohol intake can be configured using patient-reported questionnaire inputs, supporting consistent, protocol-aligned tracking.
Protocol-Aligned Rules
Configure calculation rules based on study protocol requirements.
Consistent Tracking
Standardize intake capture to support reliable review and reporting.
Study-Ready Outputs
Summaries and exports can be generated for site review workflows.
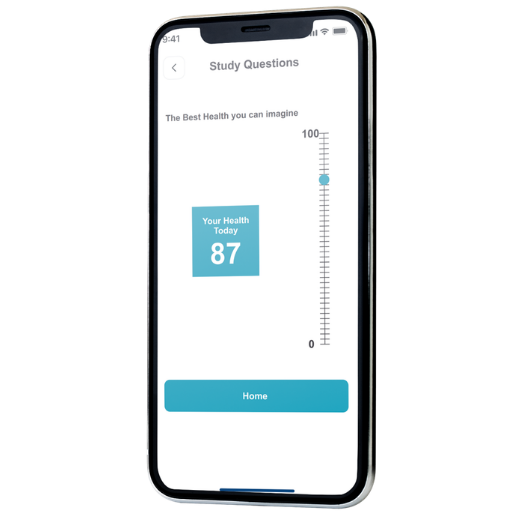
Health Score (Optional)
A configurable engagement indicator that helps teams understand daily reporting patterns and support consistent participation across study duration.
- Holistic engagement metric based on daily questionnaire submissions.
- Feedback on reported patterns to encourage positive behavior changes.
- Study-defined scoring parameters for relevance and consistency.
Advanced Features (Optional Add-Ons)
Drug Compliance
Video monitoring workflows to support medication adherence (if enabled).
Retention Tools
Engagement tools designed to support participation across trial duration.
Incident Reporting
Protocol-based incident reports and surveys for structured capture.
Scheduling & Reminders
Personalized reminders via email/text/push (configurable).
Ready to See ResearchHero eDiary in Action?
- Fast onboarding & configuration
- Training & support for sites
- Secure setup for clinical operations
